Insights from Operando and Identical Location (IL) Techniques on the Activation of Electrocatalysts for the Conversion of CO2: A Mini-Review
The paper authored by A. Dutta, K. Kiran, M. Rahaman, I. Zelocualtecatl Montiel, P. Moreno-García,
S. Vesztergom,
J. Drnec, M. Oezaslan and P. Broekmann
is published in Chimia (2021, vol. 75, pp. 733–743).
Abstract:
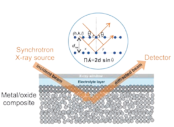
In this mini-review we compare two prototypical metal foam electrocatalysts applied to the transformation of CO2 into value-added products (e.g. alcohols on Cu foams and formate on Bi foams). A substantial improvement in the catalyst performance is typically achieved through thermal annealing of the as-deposited foam materials, followed by the electro-reduction of the pre-formed oxidic precursors prior or during the actual CO2 electrolysis. Utilizing highly insightful and sensitive complementary operando analytical techniques (XAS, XRD, and Raman spectroscopy) we demonstrate that this catalyst pre-activation process is entirely accomplished in case of the oxidized Cu foams prior to the formation of hydrocarbons and alcohols from the CO2. The actually active catalyst is therefore the metallic Cu derived from the precursor by means of oxide electro-reduction. Conversely, in their oxidic form, the Cu-based foam catalysts are inactive towards the CO2 reduction reaction (denoted ec-CO2 RR). Oxidized Bi foams can be regarded as an excellent counter example to the above-mentioned Cu case as both metallic and the thermally derived oxidic Bi foams are highly active towards ec-CO2 RR (formate production). Indeed, operando Raman spectroscopy reveals that CO2 electrolysis occurs upon its embedment into the oxidic Bi2O3 foam precursor, which itself undergoes partial transformation into an active sub-carbonate phase. The potential-dependent transition of sub-carbonates/oxides into the corresponding metallic Bi foam dictates the characteristic changes of the ec-CO2 RR pathway. Identical location (IL) microscopic inspection of the catalyst materials, e.g. by means of scanning electron microscopy, demonstrates substantial morphological alterations on the nm length scale on the material surface as consequence of the sub-carbonate formation and the potential-driven oxide reduction into the metallic Bi foam. The foam morphology on a mesoscopic length scale (macroporosity) remains, by contrast, fully unaffected by these phase transitions.
